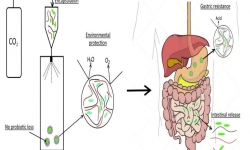
Encapsulation with Supercritical CO₂
The Technology:
Environmentally friendly process for encapsulation and micronisation:
- Supercritical CO₂ processes: Benign and environmentally friendly “green” processes using low temperatures & no organic solvents for materials processing
- Encapsulation: Imbedding an active ingredient in a carrier/shell that results in improved therapeutic benefit, controlled release, prolonged shelf-life, and/or lower dosage requirements
- Micronisation: Reduction of the average particle size of a solid material to within the micron size range to enhance functional characteristics or to unlock new applications
The unique properties of supercritical CO₂ make it ideal for the encapsulation of labile actives (probiotics, proteins, phytogenics, vitamins, essential oils etc.) or to obtain unique physical characteristics of micronized materials not possible with other processes.
What we offer:
- Platform encapsulation technology:
We have developed a novel, low-cost and scalable encapsulation technology that has shown to significantly enhance shelf-stability and gastric resistance of various probiotic strains. This technology has the potential to be applied to various other labile actives for both human and animal health.
- Customised encapsulation technologies:
Should our platform encapsulation technology not meet the client needs, we can assist in the development of a customised encapsulation technology.
- Micronisation of pure compounds:
We have the skills and expertise to micronise and engineer physical particle characteristics for a range of materials. Typically, these are: lipids, fats, waxes & polymers.
- Lab to pilot-scale development:
We assist in taking an idea from proof-of-concept, through development and optimisation, to pilot-scale manufacturing.
- Batch/Toll manufacturing:
Our pilot supercritical CO₂ facility has a capacity to produce batches of encapsulated active or micronised materials for field trials or market testing (±8 – 24kg/day) or can be used for toll manufacturing purposes, allowing the client to establish a market footprint without the need to invest CAPEX.
Our facilities:
- R&D capabilities:
Our research team has a combined +40 years’ experience in the development of encapsulation technologies/micronisation using the supercritical CO₂ process. Our research is supported by a wide range of analytical equipment and a laboratory scale supercritical CO₂ unit.
- Pilot Supercritical CO₂ Facility:
This facility will be the 1st of its kind in Africa and can be used for scalability studies, production of batch quantities or toll manufacturing (up to ±64kg/day) for commercial purposes. It can run in different modes (batch, semi-continuous and continuous)
- Quality:
Our laboratories have ISO 9001:2015 certification while the Pilot Supercritical CO₂ Facility is designed according to Hazard Analysis and Critical Control Point (HACCP) food safety specifications. Once commissioned, HACCP certification will be sought.
Knowledge Transfer Management:
The CSIR employs various knowledge transfer models:
- Contract Research:
Employed when an external client pays the CSIR at full-cost of R&D to solve a particular problem or develop their idea.
- Co-development:
When the CSIR and an external client engage in co-developing a technology. This can be a joint development on IP creation or development of existing IP.
- Toll manufacturing:
When a client pays the CSIR to produce a product according to client’s specification. Only in areas where the CSIR has the capacity.
Licensing:
The CSIR negotiates various licensing options with suitable partners within several areas: Type of Licence, Business Model, License Fees, IP Maintenance and Improvements
Other transactions:
Apart from licensing, other transactions the CSIR might consider are: equity stake in a start-up or SMME; establishing a CSIR enterprise; sale of CSIR IP.
Technical Specifications
Our pilot supercritical CO₂ facility has a capacity to produce batches of encapsulated active or micronised materials for field trials or market testing (±8 – 24kg/day) or can be used for toll manufacturing purposes, allowing the client to establish a market footprint without the need to invest CAPEX.