Advanced Search
Search Results
CSIR congratulates the Chairperson of its Board, Prof. Thokozani Majozi, for being awarded the National Order of Mapungubwe. This is the highest honour in South Africa, granted by the President of the country, and it recognises South Africans who have accomplished excellence and exceptional achievement in international research areas that have benefitted the country.
The CSIR is overseeing the design and construction of South Africa’s first purpose-designed science centre in Cofimvaba in the Eastern Cape.
The CSIR affirms that upholding the highest ethical standards in a research community is a principle of integrity, trust, collegiality and justice.
The following regulatory codes are used for research ethics background and understanding:
NHREC Ethics Guidelines - 2015
Guidelines on Ethics in Medical Research: General Principles (MRC) - Ethics book1
Guidelines on Ethics in Reproductive Biology and Genetic Research (MRC) - Ethics book 2
Guidelines on Ethics in the Use of Animals in Research (MRC) - Ethics book 3
Guidelines on Ethics in the Use of Biohazards and Radiation (MRC) - Ethics book 4
Guidelines on Ethics in HIV Vaccine Trials (MRC) - Ethics book 5
Please select the form or template relevant to your application below.
New applications
Application form for ethics approval for research: Human participants
Application form for ethics approval for research: Animal Studies
Application form for ethics approval for research: Biomedical Research
Application form for ethics approval for research: Genetically modified Organisms (GMO)
Monitoring of approved research studies:
Extension of ethical clearance/modification of approved protocol template
CSIR REC review submission process and submission dates overview
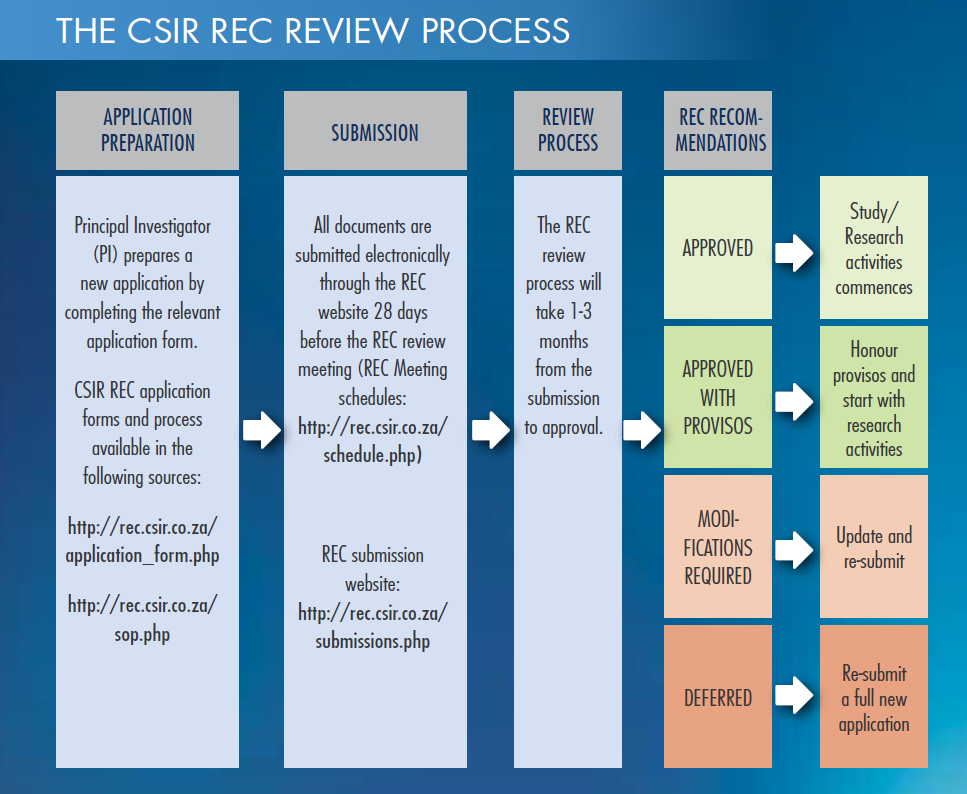
- The application form should be completed in plain English understandable to lay persons, and all abbreviations should be explained at their first time of mention.
- All sections in the application forms give a good guideline to the information required for a thorough ethical review to take place. It is therefore important that the application forms are completed in full.
- Please expand the boxes on the form to accommodate your answers.
- Please attach appendices as requested in the checklist. Where appendices required are not relevant to your study, simply mark sections accordingly.
| Research Ethics committee meeting schedule and submissions for 2019/2020 | ||
| Submission deadlines | REC review meeting dates | |
| 1st meeting | 25 January 2019 | 21 February 2019 |
| 2nd meeting | 08 March 2019 | 11 April 2019 |
| 3rd meeting | 03 May 2019 | 06 June 2019 |
| 4th meeting | 25 June 2019 | 25 July 2019 |
| 5th meeting | 16 August 2019 | 19 September 2019 |
| 6th meeting | 18 October 2019 | 21 November 2019 |
| 7th meeting | 24 January 2020 | 20 February 2020 |
Electronic submission of the application form should be made to the REC Secretariat: http://rec.csir.co.za/submissions.php
In 2019, the World Summit on the Information Society (WSIS) forum, organised and hosted by the International Telecommunications Union (ITU) in Geneva, Switzerland, celebrated its 10th anniversary. The CSIR’s Dr Jabu Mtsweni served as one of 14 high-level track facilitators, having been nominated by the Institute of Electrical and Electronics Engineers.
The Council for Scientific and Industrial Research (CSIR) is working closely with private sector and innovation partners in developing and localising technologies to support the advancement of industries in South Africa.
Media invite
The Chairperson of the Board of CSIR, Prof Thokozani Majozi, together with the CEO, Dr Thulani Dlamini, held a media briefing to provide an update on some of the latest strategic developments in the organisation, including new key research projects.
The mandate of the CSIR requires the organisation to undertake directed scientific and industrial development research to improving the quality of life of the people of South Africa.
A novel research study by the CSIR and Stellenbosch University to quantify water uses nationwide shows that, while South Africa’s largest water user is the agricultural sector, in Gauteng; industri


