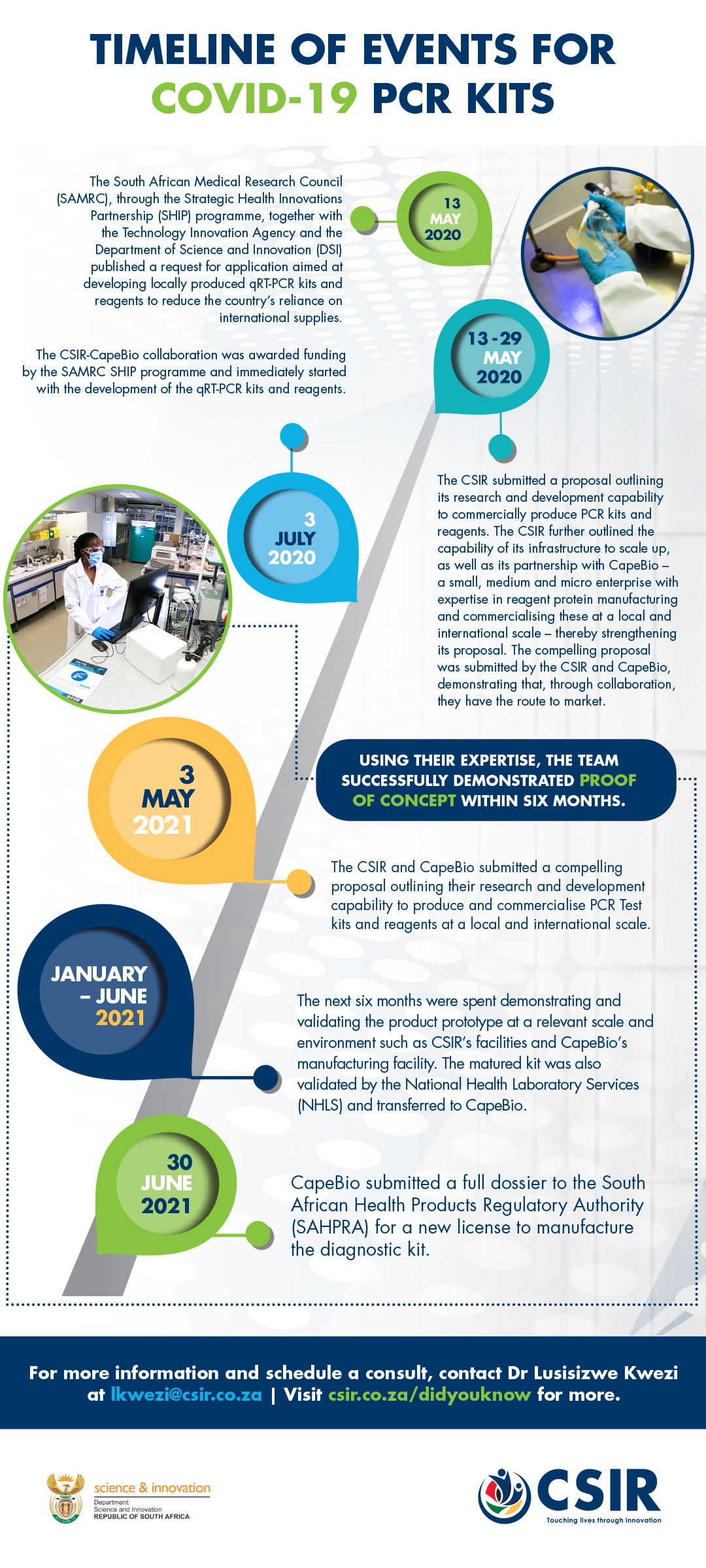
SARS-COV-2 diagnostic PCR test kit
CSIR and CapeBio lead development of local SARS-COV-2 diagnostic PCR test kit
An overburdened demand, coupled with a worldwide shortage of rapid Covid-19 test kits, prompted the CSIR and CapeBio to demonstrate their expertise in biomanufacturing as a solution to provide the country, and the region, with a locally developed PCR test kit for Covid-19.
In early 2020, the CSIR, in collaboration with CapeBio, demonstrated the efficiency of the biomanufacturing process for two enzymes, which were combined to formulate a locally produced one-step Covid-19 diagnostic assay.
The diagnostic assay has been validated in a clinical setting and proven to have the capability to detect coronavirus 2 (SARS CoV-2)-specific genetic biomarkers. Furthermore, the single-step format of the diagnostic assay reduces the turnaround time of tests and assists in managing and monitoring the spread of SARS CoV-2.
Additionally, external evaluation by the National Health Laboratory Service was passed, and the South African Health Products Regulatory Authority subsequently licensed Cape Bio to manufacture the diagnostic kit at their facility in Centurion.
This significant milestone, which took place in under a year, resulted in the kit hitting the local market in August 2021. According to CapeBio CEO, Daniel Ndima, at full operational capacity, the company is able to produce up to 5 000 kits a day, with each kit providing 1 000 tests.
This is a clear demonstration that the CSIR, through its industrialisation strategy and commitment to assisting new and emerging small, medium and micro enterprises, is touching lives through innovative biomanufacturing processes.
The research and development funded by the South African Medical Research Council and the Technology Innovation Agency enables a faster response in terms of active case identification, quarantine and contact tracing. Additionally, the localisation of the production of these reagents continues to increase accessibility to locally produced diagnostic kits – a very significant milestone that will reduce the country’s reliance on international suppliers.